 Schematic showing the electric potential generated from virus film upon heating. Heat denatures, or melts, the protein coating on the engineered phage, causing a difference in electrical potential. (Image courtesy of Seung-Wuk Lee)
Schematic showing the electric potential generated from virus film upon heating. Heat denatures, or melts, the protein coating on the engineered phage, causing a difference in electrical potential. (Image courtesy of Seung-Wuk Lee)Researchers demonstrate heat-induced pyroelectricity in viruses
Viruses are often associated with disease, but many viruses are benign and even helpful, like bacteriophages that eat harmful, antibiotic-resistant bacteria. Now, Berkeley researchers have reported that engineered viruses also can generate electricity when exposed to heat, a finding that may pave the way for next-generation biosensors and diagnostic tools.
As reported in the journal Advanced Materials, researchers discovered for the first time “heat-induced electrical potential generation on a virus,” a phenomenon known as pyroelectricity. This work may shed light on how biomaterials — cells, tissues and proteins — generate electricity at a molecular level as well as lead to the development of biomaterials with novel medical, pharmaceutical, environmental and energy applications.
“Through this study, we gained valuable insights into the relationship between protein structures and electrical properties,” said principal investigator Seung-Wuk Lee, professor of bioengineering at UC Berkeley and faculty scientist at Lawrence Berkeley National Laboratory. “We discovered that when we apply heat to virus particles, they undergo structural changes that lead to changes in spontaneous polarization and the generation of electric potential.”
Interest in bioelectricity — electric potential generated within living cells — dates to ancient Egypt and exploded in the 18th century, when Luigi Galvani discovered that electrical stimulation could induce muscle contraction in frog legs. While our scientific understanding of this phenomenon has vastly improved over the years, questions remain due to the complexity of biomaterials and the limited tools available to study them. Lee thinks viruses might help us find the answers.
“Viruses possess some remarkable properties that make them an ideal model system for studying the links between structure and function in the generation of bioelectricity,” said Lee. “These include the ability to quickly self-replicate at a massive scale, evolve and — because of their identical shape and material — self-assemble like chopsticks in a box.”
Lee and his team first demonstrated the bioelectric potential of the engineered M13 virus more than 10 years ago. Researchers reported on the piezoelectric properties of these viruses and their ability to generate electricity in response to pressure.
By genetically engineering the phage’s external protein structures, the researchers discovered that they could enhance its piezoelectric performance. And while M13 viruses lack the direct ability to sense heat, the researchers had observed that they undergo structural changes in response to thermal stimuli. These characteristics led them to suspect that the phages might possess pyroelectric properties.
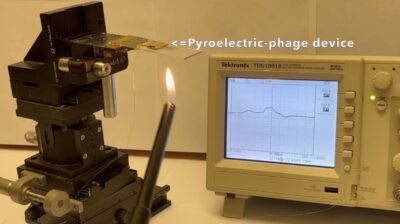
Photograph showing the electric potential generated from virus film upon heating. (Image courtesy of Seung-Wuk Lee)
In this latest study, Lee and his team put this theory to the test. They genetically engineered a portion of the M13 phage’s protein coating to create upright structures, pointing in the same direction, and in a nanometer scale.
They then observed and measured the virus’ electric response to heat. According to Lee, their findings showed that heat denatures, or melts, the protein coating on the engineered phage, causing a difference in electrical potential.
Next, the researchers investigated the M13 phage’s pyroelectric responses to other molecules. Peptides genetically tuned to be responsive to specific nonvolatile and volatile organic compounds were attached to the protein coating of the engineered pyroelectric phages. When these phages were exposed to the different solvents, they generated different pyroelectric responses depending on chemical species and their concentrations.
“Our findings showed that, depending on the chemical receptor, we could easily detect a harmless or a toxic chemical, like xylene,” said Lee.
Lee and his team believe that this approach to engineering phages with tailored pyroelectric properties could open the door to other applications. This includes building handheld devices that use pyroelectricity to sense other viruses and detect new strains.
“We envision many possibilities for this technology,” said Lee. “It may help pave the way for creating new bioelectric materials for use in bioelectronics and energy harvesting and in novel bio-inspired devices.”
Han Kim, lead author of this study, is a graduate student researcher in Lee’s lab. Co-authors include undergraduate student researchers Kento Okada and Yoonji Kwon, and postdoctoral researchers Inseok Chae and Butaek Lim.
For more information:
Virus films generate electricity when heated (video)
